Elements compounds mixtures
Elements compounds mixtures
The following text is used only for teaching, research, scholarship, educational use and informative purpose following the fair use principles.
We thank the authors of the texts and the source web site that give us the opportunity to share their knowledge
Chemistry
Elements compounds mixtures
Elements, Compounds & Mixtures Key Notes
Elements, Compounds & Mixtures
There are over 150 types of atom. A substance which contains just one type of atom is known as an element. An element is made up of one type of atom
An atom is the smallest part of an element you can get - elements consist of just one type of atom (but there are countless numbers of atoms, not just one)!
Each element has its own symbol
The atoms of different elements can join together in chemical reactions, forming compounds
The properties of compounds are usually very different from the properties of the elements they contain
A compound is two or more elements which are chemically bound together.
A mixture is made from different substances which are not chemically bound.
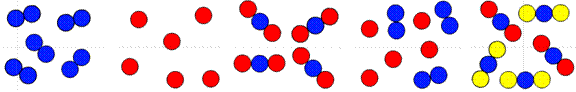

Element Element Compound Mixture Mixture
The Periodic Table
Every element has its own chemical symbol. It is usually one or two letters long (but can sometimes be three). Every symbol begins with a capital. The second and third letters are lower case
Sometimes the symbols are really obvious: O = oxygen; Li = lithium; Mg = magnesium
However sometimes it is not easy to tell what the symbol stands for (because the symbol come from a name that is not an English word) e.g. W stands for tungsten (from the word wolfram); Na for sodium (from natrium).
The chemical symbols are used all over the world, not matter what language people speak.
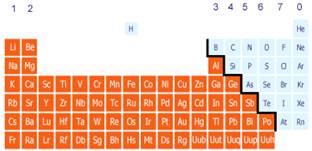

All the different elements are arranged in a chart - the periodic table
The elements are arranged so similar elements are found in the vertical columns, called groups. The horizontal rows are called periods
The metals are on the left, the non-metals on the right
Each element has its name and symbol, e.g. oxygen (O), but it also has a number - e.g. Oxygen = 8
This number is the atomic number - it tells you what element is what - e.g. oxygen always has an atomic number of 8, magnesium always has an atomic number of 12
You’ll see in the table that this number gets bigger as you go along it
Elements Combining
To make your chemical name, you simply combine the names of the elements you reacted together to make your compounds
E.g.
carbon + oxygen ® carbon oxide
sodium + chlorine ® sodium chloride
iron + oxygen ® iron oxide (rust)
copper + oxygen ® copper oxide
The ide is added to the end to tell us the elements have joined together, forming a compound
When you add oxygen and 2 other elements, the name usually ends in ate
A chemical formula is made up of the symbols for the elements it contains
E.g. carbon monoxide is CO
This tells us it contains carbon and oxygen - for every one carbon atom there is one oxygen atom. Many chemical formulas also contain numbers - these tell you what the ratio of elements in the compound is
E.g. carbon dioxide is CO2
This means that for every carbon atom there is, there are two oxygen atoms
Chemical Formula
Chemical equations show us our reactants and products
reactants ® products
They can be word or symbol equations, e.g.
carbon + oxygen ® carbon monoxide
2C + O2 ® 2CO
Elements and compounds can be represented using chemical formula. We use numbers in the formula, which represents the number of atoms present (as a ratio)
C = 1 atom of carbon (C)
O2 = 2 atoms of oxygen (O)
CO2 = 1 atom of carbon (C); 2 atoms of oxygen (O)
NaCl = 1 atom of sodium (Na); 1 atom of chlorine (Cl)
H2SO4 = 2 atoms of hydrogen (H); 1 atom of sulphur (S); 4 atoms of oxygen (O)
Separating Mixtures
To separate mixtures we can use a variety of techniques - the method used depends upon the mixture: -
- Filtration
- Evaporation
- Distillation
- Chromatography
Filtration separates an insoluble solid (does not dissolve) from a liquid, e.g. sand and water can be separated using filtration
Evaporation separates a soluble solid (dissolved) from a liquid, e.g. water can be evaporated from salty water, leaving the salt behind
Distillation can separate a liquid from a solution. The liquid evaporates from the solution, is cooled and condenses (which you then collect). Different liquids boil at different temperatures, so it is possible to distill a solution containing many liquids, e.g. water can be separated from salty water
Chromatography can separate dissolved substances which have different colours. It works because some of the coloured substances dissolve in liquid better than others, so traveling further up the paper, e.g. an ink or dye can be split into the different colours which make it up
Melting & Boiling Points
Substances change states at given temperatures. This is very useful information to know, e.g. a mercury thermometer will not work below -39°C. Below this temperature the mercury will freeze (become solid), so cannot move up or down the thermometer!
Melting point - this is the temperature a substance turns from a solid to a liquid. Different substances have different melting points, e.g. water melts at 0°C; ethanol melts at -114°C
There is a fixed temperature where a substance turns from a solid to a liquid. Don’t forget, this point is also the temperature where a substance turns from a liquid to solid (its freezing point), i.e - water melts at 0°C and above (solid to liquid); and it freezes at 0°C and below (liquid to solid)!
Boiling point - This is the temperature at which a substance evaporates as quickly as it can (liquid to gas). Different substances have different boiling points, e.g. water boils at 100°C; ethanol boils at 78°C
There is a fixed temperature where a substance boils from a liquid to a gas. Don’t forget, this point is also the temperature where a substance turns from a gas to liquid (its condensing point), i.e - water boils at 100°C and above (liquid to gas); and it condenses at 100°C and below (gas to liquid)!
You can identify the state of a substance if you know its temperature, melting and boiling point:
- If the temp. is below the melting point, the substance will be a solid
- If the temp. is between the melting and boiling point, the substance will be a liquid
- If the temp. is above the boiling point, the substance will be a gas
If you add other substances then the temperature your original substance freezes, melts and boils can change, e.g. salt is added to water to lower the melting point (and freezing point). This is why salt is spread on roads during winter
Normally the water would freeze at 0°C. Adding the salt means the temperature must be even colder than 0°C for the ice to freeze - useful for cold nights!
Source : http://web.me.com/davecrowley/Noadswood_Science/Revision_-_Year_8_-_Elements_files/Elements,%20Compounds%20%26%20Mixtures%20Key%20Notes.doc
Web site link: http://web.me.com/davecrowley/Noadswood_Science/
Author : not indicated on the source document of the above text
If you are the author of the text above and you not agree to share your knowledge for teaching, research, scholarship (for fair use as indicated in the United States copyrigh low) please send us an e-mail and we will remove your text quickly.
Syllabus
OC3 Understand what an element is and recall that all known elements are listed in the Periodic Table; understand what a compound is and what a mixture is; recall that when elements combine to form compounds they may lose their individual properties
OC4 Examine a variety of substances and classify these as elements or compounds (using the Periodic Table as a reference)
OC12 Compare the properties of the simple compounds H2O, CO2, MgO and FeS to those of the constituent elements
OC13 Compare mixtures and compounds made from the same constituents, and understand that an alloy is a mixture
Student Notes
We saw in the last chapter (States of Matter) that everything is made of atoms.
It turns out that there are many different types of atom (over 100 in fact), and some substances are made from just one type of atom, while other substances are made from combinations of different types of atom.
Elements
The different types of atom are arranged in terms of their size in a table called the Periodic Table of elements.
Elements are substances which are made up of only one type of atom
Molecules
Some atoms form a very strong attachment (‘bond’) to another atom or atoms and as a result they always go around in groups of two or more atoms. We call these guys ‘molecules’.
It is very hard to break these ‘bonds’ and we say that in this case the atoms are ‘chemically combined’. We will look at this bonding in detail in another chapter later.
Basically a molecule is like a very small group of atoms that go around together (they are still too small to see).
Examples:
For the element oxygen the atoms go around in pairs of oxygen atoms.
These are called oxygen molecules and that is why oxygen is often represented as O2.
For the compound water the atoms go around in ‘gangs’ consisting of two hydrogen atoms and one oxygen atom. These ‘gangs’ are called water molecules and that is why water is often represented as H2O.
Compounds
A compound is formed when two or more atoms of different elements combine together chemically
My head hurts; what’s the difference between a compound and a molecule?
All compounds are molecules, but not all molecules are compounds (e.g. H2 is a molecule because it is composed of two atoms chemically combined, but because they are both hydrogen atoms the molecule is not a compound).
Remember when we said that atoms are so small that you can’t see them?
Well molecules are made up of small groups of atoms so you won’t be able to see them either.
A compound however might be something like table-salt; you can hold it in your hand. It is a compound because it is made up of two different types of atom – in this case sodium and chlorine. The chemical name for table-salt is NaCl.
The interesting thing is that both sodium and chlorine can be fatal if ingested (swallowed) but when the two go together to form table salt the result is perfectly safe (once you don’t eat too much!).
We can summarise this as follows:
When elements combine to form compounds they may lose their individual properties.
Other examples of compounds and their constituent elements
Compound |
Symbol |
State of matter |
Elements in the compound |
Water |
H2O |
liquid |
Hydrogen (gas) |
Carbon dioxide |
CO2 |
gas |
Carbon (solid) |
Magnesium oxide |
MgO |
solid |
Magnesium (solid) |
Iron sulphide |
FeS |
solid |
Iron (solid) |
Mixtures
If a substance is made up of different components but they are just mingled together rather than combined at an atomic level then we call this a mixture.
A mixture contains two or more different substances mingled together but not chemically combined
Mixtures can be (fairly) easily separated, whereas compounds cannot be easily separated.
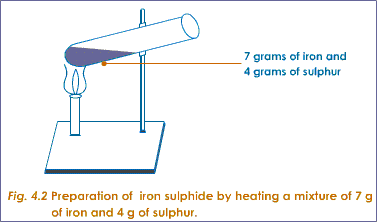
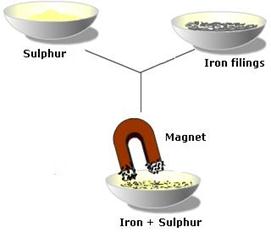
For example if you pour sulphur power over iron filings and mix them together you get a mixture, and to separate them simply use a magnet which will attract the iron filings and leave the sulphur powder behind.
However if you heated the mixture to a very high temperature their chemical compositions will change and the two elements will ‘bond’ together chemically to become a new substance called iron sulphide. This cannot now be separated and the result is called a compound.
Exam Questions
- [2006 OL]
Table Salt |
Identify one mixture and one compound from the list on the right.
Element |
- [2009 OL]
Complete the following sentence using the words from the list on the right.
Water is an example of a _______________ and hydrogen is an ________________ found in water.
- [2007 OL]
Write the name of each of the two elements present in water.
Aluminium |
Copper |
- [2009 OL]
What is the symbol of each of the metallic elements on the right?
- [2007]
Marie Curie showed the existence of the element radium and she produced 0.1 g of the compound radium chloride in 1902 by processing tons of pitchblende ore obtained from mines in Bohemia.
Explain the underlined terms.
- [2010]
Give two different properties of the element magnesium compared to the compound magnesium oxide.
Exam Solutions
MIXTURE |
COMPOUND |
Air |
Table salt |
- Water is an example of a compound and hydrogen is an element found in water.
- Hydrogen and oxygen
- Al
- Cu
- Element: An element cannot be broken down into simpler substances.
Compound: A compound is composed of two or more elements chemically combined.
- Magnesium: is ‘silver’/ metal/ shines/ bends/ ductile/ malleable/ reacts with acids releasing hydrogen/ conducts electricity/ conducts heat/ burns in air (Oxygen)…
Magnesium Oxide: white/ powder/ base/ does not burn/ does not conduct electricity/ does not conduct heat…
Other Test Questions
- What is an element?
- What is a compound?
- Give four examples of compounds.
- What is a molecule?
- What is a mixture?
- Explain the difference between a physical change and a chemical change in chemistry:
Physical:
Chemical:
- Give one example of a physical change and a chemical change that you have observed
Physical:
Chemical:
- A student carried out an investigation comparing the properties of a mixture of iron and sulphur and the compound iron sulphide.
Name and explain a method that the student could use to separate the iron from the sulphur in the iron and sulphur mixture.
Method of separation:
Explanation:
Could the method of separation that you have suggested be used to separate the iron from the sulphur in the compound iron sulphide?
Explain your answer.
- Which two of the following five examples represent a chemical change?
- Rusting of iron
- Melting of ice
- Burning of gas
- Grinding of coffee beans
- Magnetising of steel
Source : http://www.thephysicsteacher.ie/JC%20Science/JC%20Chemistry/Student%20Notes/2.%20Elements,%20Compounds%20and%20Mixtures.doc
Web site link: http://www.thephysicsteacher.ie/
Author : not indicated on the source document of the above text
If you are the author of the text above and you not agree to share your knowledge for teaching, research, scholarship (for fair use as indicated in the United States copyrigh low) please send us an e-mail and we will remove your text quickly.
Elements compounds mixtures
Elements compounds mixtures
Elements compounds mixtures
This is the right place where find the answers to your questions like :
What is Elements compounds mixtures? What does Elements compounds mixtures mean ? Which is the meaning of Elements compounds mixtures?
Elements compounds mixtures chemistry notes
Alanpedia.com from 1998 year by year new sites and innovations
Main page - Disclaimer - Contact us